This is a scientific article that I had written for one of my reports in my undergraduate years. I thought of sharing it here for the people with a scientific and medical background.
INTRODUCTION
A tumor is an abnormal proliferation of any cells in any part of the body. It can be benign or malignant which invades surrounding tissue or spreads through the lymphatic system, to other parts of the body. The ability to spread is called metastasis. Hence, cancer can be defined as a malignant tumor possessing the ability to invade and metastasize (GM, 2000)
There are multiple causes of cancer, some of them being chemicals, ionizing radiations, viruses, hormonal changes, reduced ability to repair DNA. These carcinogens, damage DNA by inducing mutations (GM, 2000). Cancer development is complex process involving multiple steps like tumour initiation(through genetic changes), tumor progression(further secondary mutations) and clonal selection(formation of a persistent cell clone).
Cancer cells have defects in the cell cycle checkpoint genes and hence current therapies like radiation therapy, chemotherapy, surgery focus on damaging DNA and cell machinery which unfortunately destroys normal cells along with cancer cells. These usually fail to remove all cancer cells from the body leading to their resistance against the treatments (Alberts, et al., 2002). Although understanding the exact biology of this disease can help in development of future effective treatments.
Introduction to Malignant Melanoma
The malignancy of melanin producing cells, melanocytes, is called malignant melanoma (MM). These cells are primarily found in the skin but are also found in the gastrointestinal tract, eyes, ears, genital and oral mucosa and the leptomeninges (McCourt, et al., 2014).This essay focuses on cutaneous malignant melanoma(CMM) which is the melanoma of the skin. This is the most common and aggressive form of skin cancer (Alonso, et al., 2004).
It was considered a rare cancer historically although its incidence has risen faster than any of the cancers in the last fifty years (Matthews, et al., 2017). About 87,110 people in the United States alone have been predicted to be diagnosed with MM in 2017. Fair skinned people have decreased melanin content and hence have less protection from ultraviolet rays(both UVA and UVB) making them more susceptible to melanoma (Matthews, et al., 2017).
Even though MM forms only 5% of all cutaneous malignancies, it accounts for majority of deaths and is considered the most lethal form of skin cancer (Matthews, et al., 2017).
It presents with signs and symptoms which are called the “ABCDEs” or malignant melanoma. These are observed on the skin or the mole (McCourt, et al., 2014).
A: asymmetry
B: border irregularity
C: colour variation
D: diameter > 6 mm
E: evolving(changing)
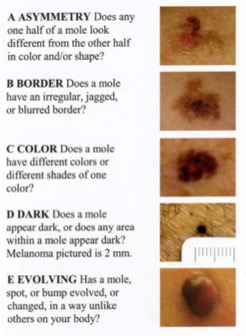
Figure 1: Signs and symptoms of melanoma (Goldsmith, 2014).
Normal Structure and Function of Skin
The skin is composed of two layers:
Epidermis– This layer originates from the ectoderm and consists primarily of melanocytes and other cells like keratinocytes, Langerhans cells and merkel cells. At the bottom of this layer, there are tactile cells which sense pressure changes.
Dermis– A deeper layer resting on a subcutaneous fat layer originates form the mesoderm. It is made up of connective tissue. It contains collagen, elastic fibers, nerves, adipocytes, blood vessels and fibroblasts.
Skin protects against ultraviolet radiations, toxins, microorganisms, trauma etc. It has multiple functions like protection, vitamin D production, thermoregulation, water homeostasis, producing hormones and growth factors. It acts as a nervous transducer and has exocrine and immunologic functions.
Main functions of melanocytes are protection from the sun and skin pigmentation. The functions of melanin are still under research but melanin might be involved in cation chelation, antioxidation and ‘free radicals sinking’ (Liu & Sheikh, 2014). Most neoplasms of melanocytes arise from the skin(either from a pre-existing lesion or de novo) (Liu & Sheikh, 2014) (Heistein & Acharya, 2017)
According to (Liu & Sheikh, 2014)
There are four main types of melanoma
- Superficial spreading: 2 cm in diameter, look flat but in the later stages they may become irregular and elevated. They have “variegated” colours, peripheral notches, indentations, or both. (Heistein & Acharya, 2017).
- Nodular: Generally blue-black but, in some circumstances, lack pigment.
- Lentigo maligna: They begin as small, freckle-like lesions; diameter being larger than 3cm, they look flat, tan, with marked notching of the borders.
- Acral lentiginous melanomas: Found on the soles, palms and look like flat, tan or irregularly bordered brown stains. Ulcerations may occur in later stages of subungual(under fingernail/toenail) lesions(brown/black) (Heistein & Acharya, 2017).
ETIOLOGY AND PATHOGENESIS:
There are three main causes are:
- Ultraviolet radiation: This is the major cause of melanoma. The UVB portion of radiation mutates the DNA by forming cyclobutene pyrimidine dimers or causes deamination of cytosine into thymidine (Sullivan & Fisher, 2014).The UVA radiations penetrate deeper into the skin but do not damage the DNA directly, instead, they produce reactive oxygen species(ROS) (Thaler, et al., 2010). ROS cause oxidative damage and double and single stranded breaks. UVA also induces the photoproduct 8-oxo-7,8oxoG and cyclobutene pyrimidine dimers(CPDs) in human skin. Basically, UVA leads to the formation of ‘recurrent or hotspot’ mutations at methylated CpG(cystine guanine in 5’ to 3’ direction) sites in the genome of an individual (Thaler, et al., 2010).
- Melanocyte nevi: These are benign neoplasms of the melanocytic lineage (Bastian, 2014).The Melanocyte nevi, commonly known as a birth mark of mole, have a large number of melanocytes leading to production of a dark spot on the skin due to high melanin content. When these nevi start to increase on the skin in later years and start to change in color, shape or texture, they become a strong indication of malignant melanoma (Liu & Sheikh, 2014).These can be removed with proper surgical excision and the chances of the person becoming cancer free are higher as compared to metastatic melanoma.
- MITF: This transcriptional regulator is given the name of the “master regulator of melanocyte development.” It is still unclear how MITF gets dysregulated leading to melanocytic transformation to malignancy but it seems to directly regulate anti-apoptotic genes, metabolic regulator PGC1α, cell cycle regulator CDK2 and various other genes. MITF is directly phosphorylated by MAPK and ubiquitin dependent proteolysis and has an active role in most melanomas. It is also linked to BRAF and NRAS oncogenes (Sullivan & Fisher, 2014).
The molecular and cellular changes associated with Melanoma
The few main signaling pathways are:
MAPK signalling
The tumor progression is dependent on cyclins(cyclin D1) and the mitogen-activated protein kinase (MAPK) pathway.
This pathway(initiated by tyrosine kinases or integrins) is the most important one in CMM cell proliferation and survival. It involves MAPK kinase(MEK), the extracellular signal-regulated protein kinase(ERK), p38 MAPK and the Jun NH2-terminal protein kinase(JNK) activation pathways. This cascade activates transcription factor AP-1 which regulates the cell growth and differentiation. MAPK inhibits apoptosis of cells mainly through regulation of various pro and anti-apoptotic genes of the B cell lymphoma 2 family members(BIM, BAD, BMF etc) (Sullivan & Fisher, 2014). The most critical effect of MAPK is inhibition of BH3-protein BIM. Activation and alterations in the MAPK pathway gives cells the ability to become malignant and invasive through the RAS/RAF/MEK/ERK pathway (Piérard, 2012)
P13K Signalling
This is the second major signalling pathway playing a role in melanoma pathogenesis. The activation of P13K is similar to MAPK activation leading to conversion of PIP2 and PIP3 which further bind and activate AKT/protein kinase B. NRAS activation, loss of neurofibromatosis 1(NF1) function, oncogenic mutation of AKT3, loss of P13K regulator or phosphatase tensin homologue can activate P13K pathway in melanoma. AKT pathway activates TORC1, 2(the mammalian target of rapamycin complexes 1 and 2) through tumour suppressor proteins TSC1, TSC2(tuberous sclerosis complex 1 and 2) which regulate mTOR.
TORC activation triggers the phosphorylation of phosphor-S6(ribosomal protein) leading to proliferation, neovascularisation and resistance to apoptosis.
Expression of TSC1 and 2 has an importance in the BRAF-mutant melanoma as BRAF is involved in the downregulation of the activator proteins(LKB1) of TSC. This shows a connection between MAPK and P13K (Sullivan & Fisher, 2014).
Cyclin-dependent kinase N2A (CDKN2A)
Studies show that the loci on chromosomes 1 p36 and 9p21 play and important role in melanoma. Within the 9p21, p16(CDKN2A) gene negatively interacts with CDK4 thereby inhibiting cell cycle. Many cancers have been linked to the mutation of CDKN2A. the locus of this gene comprises of four exons encoding for p16INK4a and p14ARF, both of which are potent tumour suppressors and play important roles in in apoptosis and cell cycle regulation.
The binding of p16INK4a to CDK4/6 inhibits and prevents it from phosphorylating the RB protein, the hyperphosphorylation of which triggers release of E2F1(transcriptional inducer of S-phase genes). Loss of p16INK4a helps in re-entry into cell cycle through G1-S transition.
p14ARF binds at the N terminus of human double minute-2(HDM2) protein promoting its rapid degradation. HDM2 ubiquitinates and brings about p53 destruction. Hence p14ARF loss causes destabilization of p53. Thus, CDKN2A mutation eliminates both p53 and RB pathways via loss of p14ARF and p16INK4a, respectively (Sullivan & Fisher, 2014).
Oncogenic Mutations
BRAF: These are mutations in the serine/threonine kinase domain and are present in almost 50% of melanomas. These are two kinds of BRAF mutations: V600E where valine at the 600th position is exchanged by glutamic acid and V600K where its exchanged by arginine. In the presence of BRAF, the engagement of RAS-RAF and RAF dimerization is not required for the activation of MEK and ERK for the further activation of MAPK pathway (Sullivan & Fisher, 2014). Some researchers suspect that these might be involved in the melanocytic neoplasm formation (Liu & Sheikh, 2014).
NRAS: These are known as neuroblastoma RAS oncogenes (Liu & Sheikh, 2014) and are found in almost 15-20 % melanoma patients. NRAS and BRAF mutations do not usually occur in the same patient. NRAS brings about oncogenic signalling through MAPK and P13K pathways (Sullivan & Fisher, 2014). These mutations present a thick tumour with vertical growth and are also involved in melanoma initiation (Liu & Sheikh, 2014).
PROGRESSION AND STAGING
Most cells show a radial and slow growth in the beginning. This stage is easily curable. In the second or later stages, the cells show a vertical growth pattern. This is the tumorigenic CMM where the cells grow rapidly and cure becomes uncertain (Piérard, 2012). Stage III and IV are when it turns completely metastatic and the cure rate rapidly declines(Alonso, et al., 2004)
There are four main staging systems
- The Clark scale: evaluates the depth of the lesion.
- The Breslow scale: evaluates the thickness of the lesion.
- TNM staging: evaluates the thickness of the lesion and its spread to lymph nodes and metastasis.
- Number staging: information of the depth of the lesion and TNM staging.
Table 1: Breslow staging system
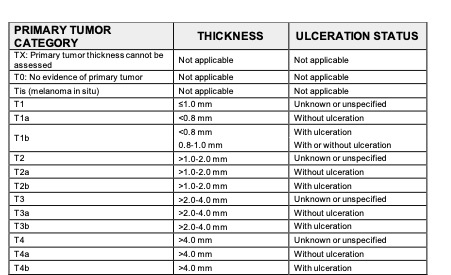
(Gershenwald, et al., 2017)
Table 2: TNM staging
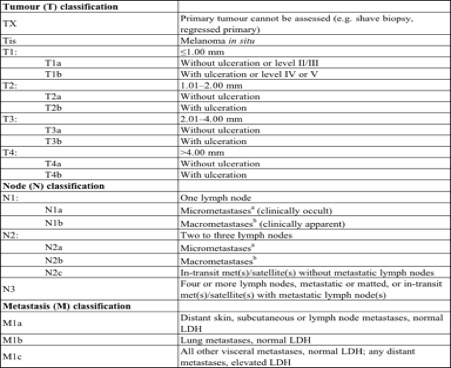
(Mohr, et al., 2009)
Table 3: TNM staging
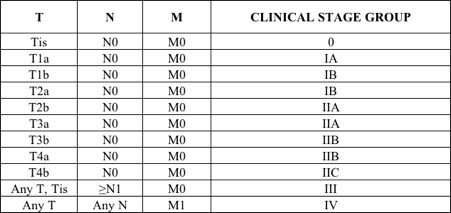
(Gershenwald, et al., 2017)
Table 4 : TNM staging
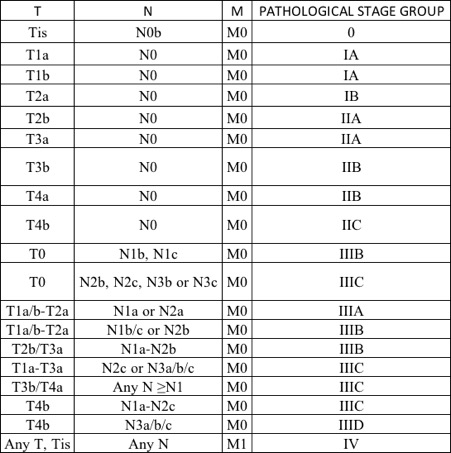
(Gershenwald, et al., 2017)
PROGNOSIS
Studies show that prognosis becomes poorer with age. Women have shown to have a better survival rate than men. This might be due to women performing self-examination and visiting dermatologists more often than men (Lideikaitė, et al., 2017). Apart from age and sex, tumor thickness, higher number of lymph nodes, anatomic site and distant metastasis are also some of the factors for prognosis (Heistein & Acharya, 2017).
| DISEASE STAGE | 5-YEAR SURVIVAL RATE |
| I | > 90% |
| II | 45% – 77% |
| III | 27% – 70% |
| IV | < 20% |
DIAGNOSIS
- The gold standard for diagnosing CMM are excisional biopsy and histological examination of suspicious lesions (C & HA, 2017).
- Another method now widely used by skin cancer multi-disciplinary team members and some doctors is dermatoscopy.
- Sequential photography for monitoring moles can also help patients with lots of moles.
- Mole mapping is another method used for examination of moles. This involves taking general photographs of the skin to map where the moles are present and can help in the future to notice new suspicious moles appearing.
- Genetic testing of melanoma tumours can help in understanding different signalling pathways involved in the development of tumour cells. Main genetic tests are done for BRAF, NRAS and c-kit mutations.
(National Collaborating Centre for Cancer (UK). , 2015)
TREATMENTS:
Surgeries like wide local excision with sentinel lymph node biopsy, elective node dissection, or both are definitive treatments for early-stage melanoma (Heistein & Acharya, 2017).
Modulation of immune system
The therapies that involve the immune system to overcome tumour mediated immune anergy initially consisted of cooley’s toxin and BCG which are non-specific immunologic agents. The recent therapies use cytokines like interferon-alpha and interleukin 2 (Sullivan & Fisher, 2014).
Dacarbazine (DITC)
DITC was the first chemotherapeutic drug approved by FDA in 1970. This works by DNA alkylation forming crosslinks in helices leading to DNA denaturation ultimately killing the cancer cell (Foletto & Haas, 2014).
Bio Chemotherapy
This refers to the combination of chemotherapy and immunotherapy which works best with the combination of DTIC, cisplatin, vinblastine, IFNα-2β, and IL-2 (Foletto & Haas, 2014).
Ipilimumab
This is a Monoclonal antibody (Mab) which has been recently approved by the FDA for the treatment of stage III, IV and recurrent melanoma.
This mab interacts with CTLA-4 (which interferes with antigen presentation and disrupts the immune system in cancer) blocking the inhibition ultimately leading to activation of the immune system again. This allows the immune system to recognise the malignant cells and destroy them (Foletto & Haas, 2014).
Vemurafenib
This is a new discovery representing a major turning-point in melanoma research. It is a BRAF V600 inhibitor which slows down the progression of the lesions when used for stage III and IV (Foletto & Haas, 2014).
Future therapies:
Bevacizumab is a Mab that inhibits vascular endothelial growth factor A blocking angiogenesis, may be useful in the treatment Stage 3 patients. Although, new studies need to confirm if it works in combination with drugs like ipilimumab (Velho, 2012).
Bortezomib which is a proteasome inhibitor has been tested in combination with some drugs in patients. Sunitinib which arrests growth and sensitises melanoma cells to bortezomib can be combined with it to provide therapeutic benefit (Velho, 2012).
famotidine and IL-2 combination can increase lymphokine-activated killer cell activity helping in tumour killing (Velho, 2012).
mTOR inhibitors such as Everolimus and temsirolimus are also under research (Velho, 2012).
CONCLUSION
Melanoma can metastasize to various body parts but its pattern and route is unpredictable. Suspicious lesions can be identified by photographic techniques. Drug therapies are proven to be more effective than surgeries or chemotherapies.The recent drugs approved by FDA have raised many hopes in treating malignant melanoma and hopefully with future therapies, individual cancer vaccines and genetically modified T cell adoptive transfer, Malignant Melanoma will become a completely curable disease (P, et al., 2017).

Woaho! I was just doing a course on cancer, and this is awesome!!!
LikeLiked by 1 person
Yaay! Glad you liked it!
LikeLiked by 1 person
Nice blog 💕
LikeLiked by 1 person
Thank you!
LikeLike
Wowww! That was quite a bit… I have a question though. Does the type of melanoma have any effect on how deadly it is?
LikeLiked by 1 person
Yes, the type definitely can affect the severity. All the types follow the TNM staging and definitely the higher the stage, the greater danger it possesses. But if we exclude the staging part, the type can have some effect on how deadly it can get. For example, nodular melanoma is the most dangerous type. Acral lentiginous melanoma which is common in African people, is hard to spot so it’s often ignored and hence it can reach the stage where it has really spread badly. So that’s how types can become deadly. Hope that helps?
LikeLiked by 1 person
Ahhhh…I’ll probably just go through it a few times to better understand. Thanks for taking the time out to reply.
LikeLike